Engineering |
Modularity and Standardisation in genetic engineering
|
The modularity and standardization of electronic components are central to the architecture of emerging electronic and digital technologies. Following engineering modularity and standardization principles, we have conceived and created a bidirectional bio-electrical communication platform by both genetic and electrical engineering. In our bio-electronic platform, not only do we apply engineering principles to biological systems, but, we go a step further by engineering bacterial populations to act as electronic components. We have designed and tested a library of parts and constructs that, when transformed into bacterial populations, behave analogously to electronic components. Building on the analogy from the world of electronics, we see our collection like the electronic components on the shelves of a hardware workshop where electronic designers can swap resistors and capacitors in a circuit. Our main Human Practices takeaway, from our interviews with Brian Ringley, Cesar Harada, and Xiao Xiao, was that to be useful and serviceable to the engineering community, our bio-electronic platform had to be as modular as possible. We have used engineering principles to achieve our engineering goal of modularity in our genetic constructs. |
What
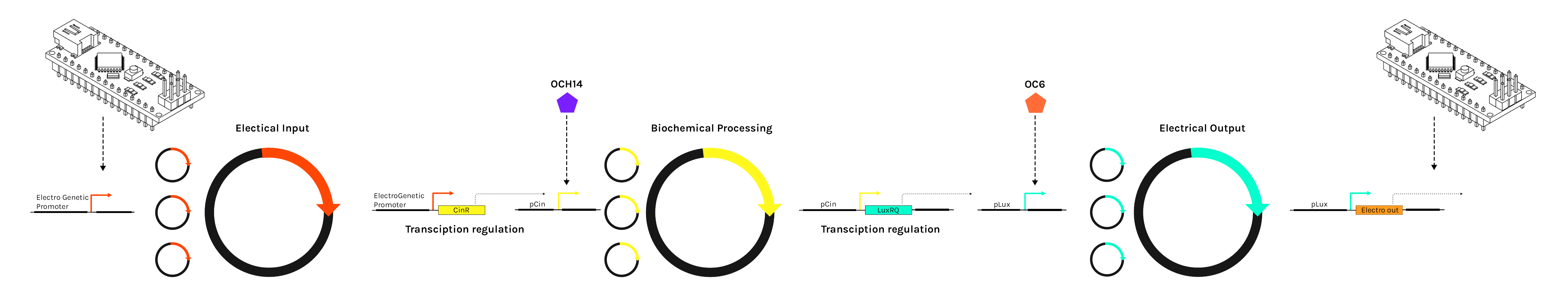
Our wetware platform is made of genetic constructs organized into three categories:
|
The modularity of our composite parts is based on two systems:
- The three Input, Processor, and Output components depend on a 3 plasmid system.
- The connection between these components is established by a standardized signal transduction communication pipeline using a tunable combination of transcription factors, promoters, and their associated chemical inducers.