Materials and methods |
|
As previous experiments [3] failed in ruling out electrical signals (as none of the signals what the HTEA was capable of gating provoke complete or major growth arrest), we turned out to a range of voltages that have been shown capable of inducing changes in membrane potential[4]. We used the HTEA in conjunction with the AC Dispatcher (ACD) and the Electro Planner to shock the bacteria with AC current of different amplitudes (from 0.5volts to 4.5 volts). Frequency and pulse durations were always respectively 100Hz and 5 seconds. All AC current waveforms were sine waves. We used the MHS2300A dual function generator to generate the sine waves and the MHS2300A Python library to communicate with the Electro Planner. The day before conducting the experiments, the cells were grown to saturation overnight in M9 minimal media (0.4% Glucose, complete Amino Acids).0 On the morning of the experiement cultures were rediuluted in fresh medium to a ratio of 1:100. Cultures were grown for 4 hours to reach mid log phase accroding to growth curved obtained in a previous experiment[5]. Experiments were done in triplicates. |
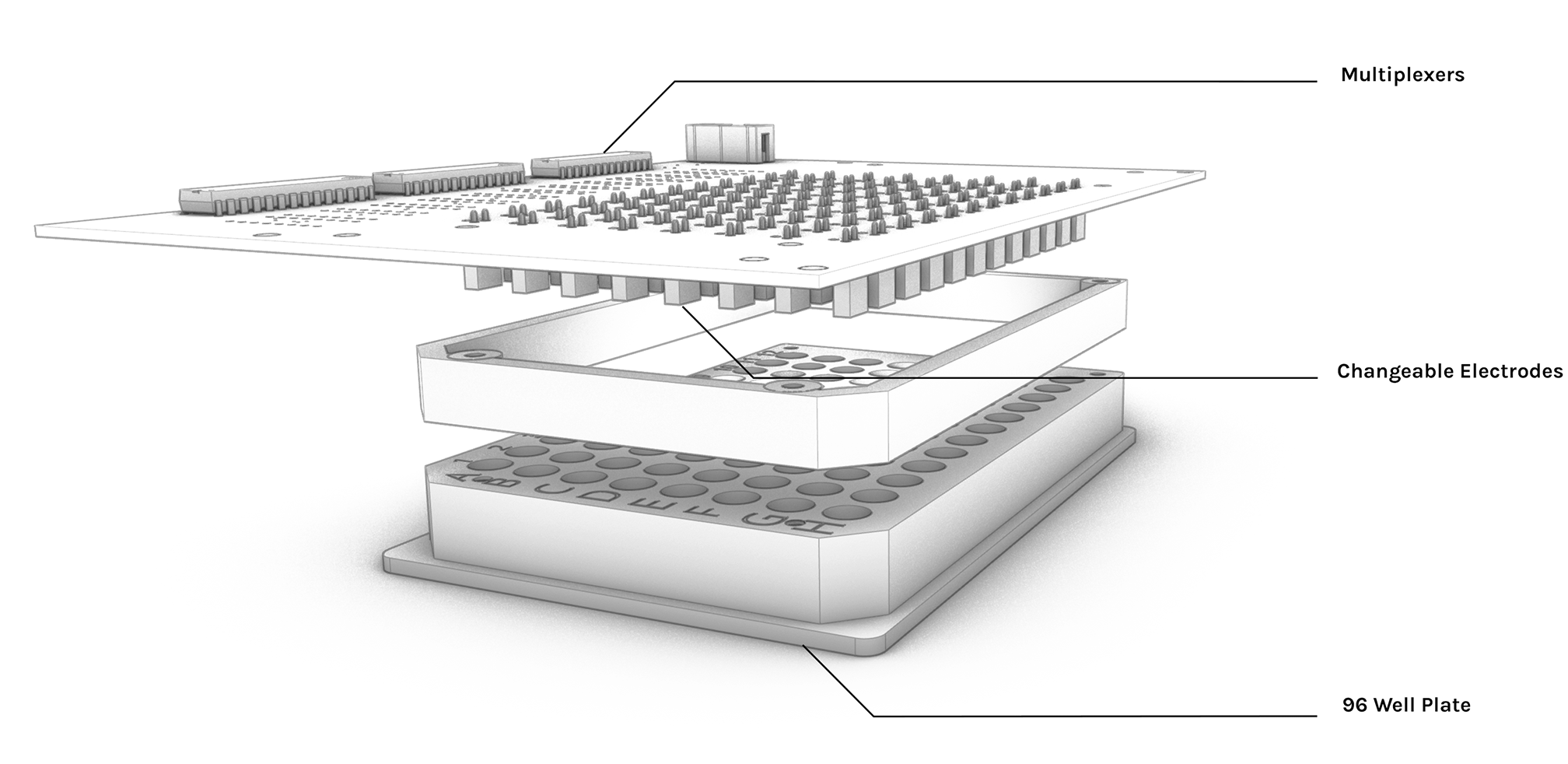
High throughput Electro Actuator (HTEA)