Introduction
Our team strove to develop eight basic parts and three composite parts. Due to limited resources and time and unprecedented challenges, our team we successful in only developing four basic parts and two composite parts. While we were unable to test them prior to the competition, we decided to document our parts so to contribute to any future projects that wish to delve into this area. For the convenience of other teams we have added Fasta files, plasmid maps, and snapgene DNA files for each part created/planned. We hope this can help others better understand our project. Since no functional characteristics were determined for any part, only descriptions of what each part does in-vivo (from the literature) will be presented; alongside what its function for our project was meant to be.
All part figures on this page were created with BioRender.com and SnapGene software.
Basic Parts: Hpa
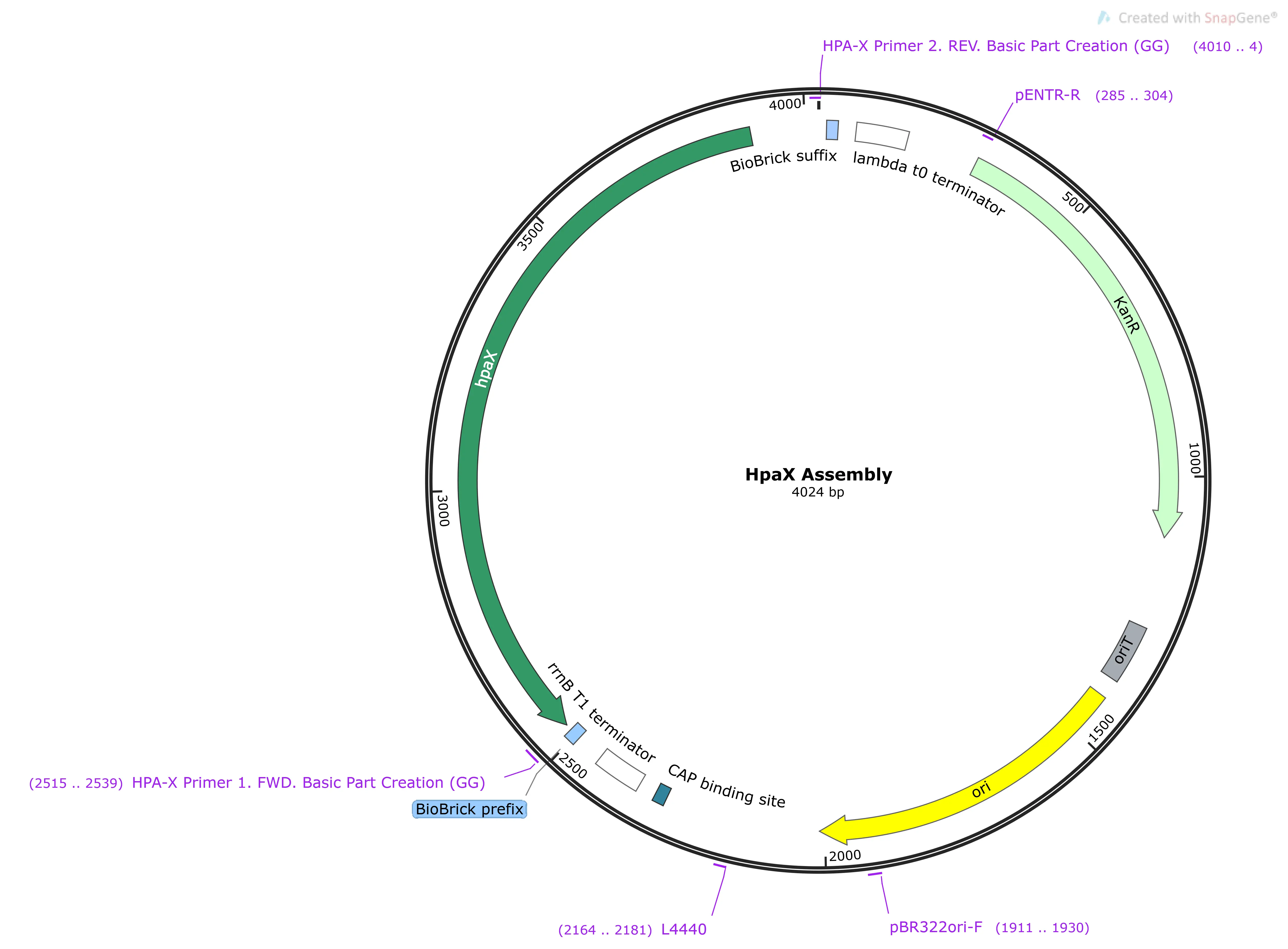
HpaX
HpaX encodes a transmembrane facilitator protein used for the import of 4-HPA.1 It was meant to act as a specific inducer of PBC-BMD expression in conjunction with HpaA.
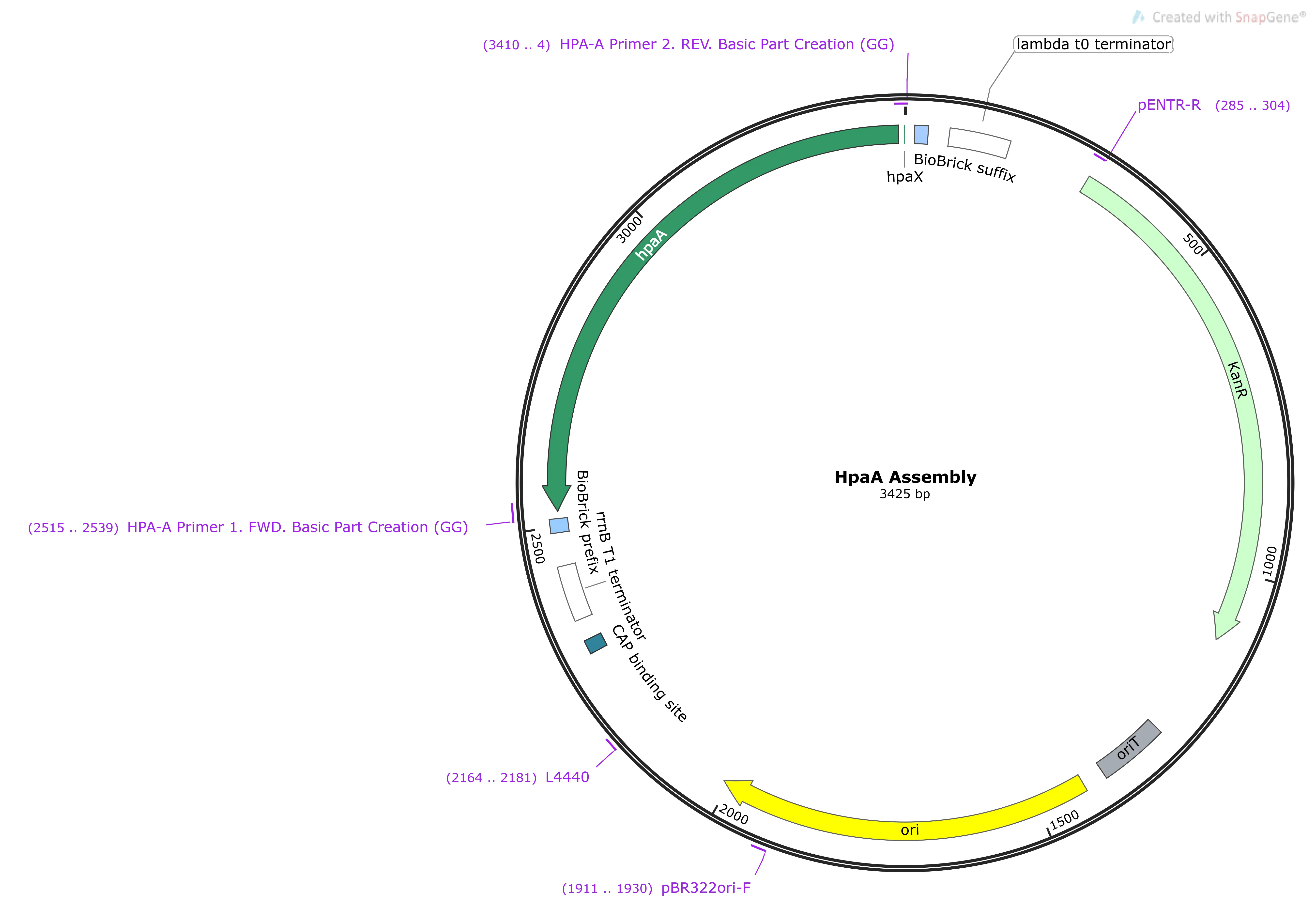
HpaA
Encodes a transcription factor which, when bound to 4-HPA, becomes active; binding to Pbc (PBC). Anything downstream of Pbc is then transcribed.2
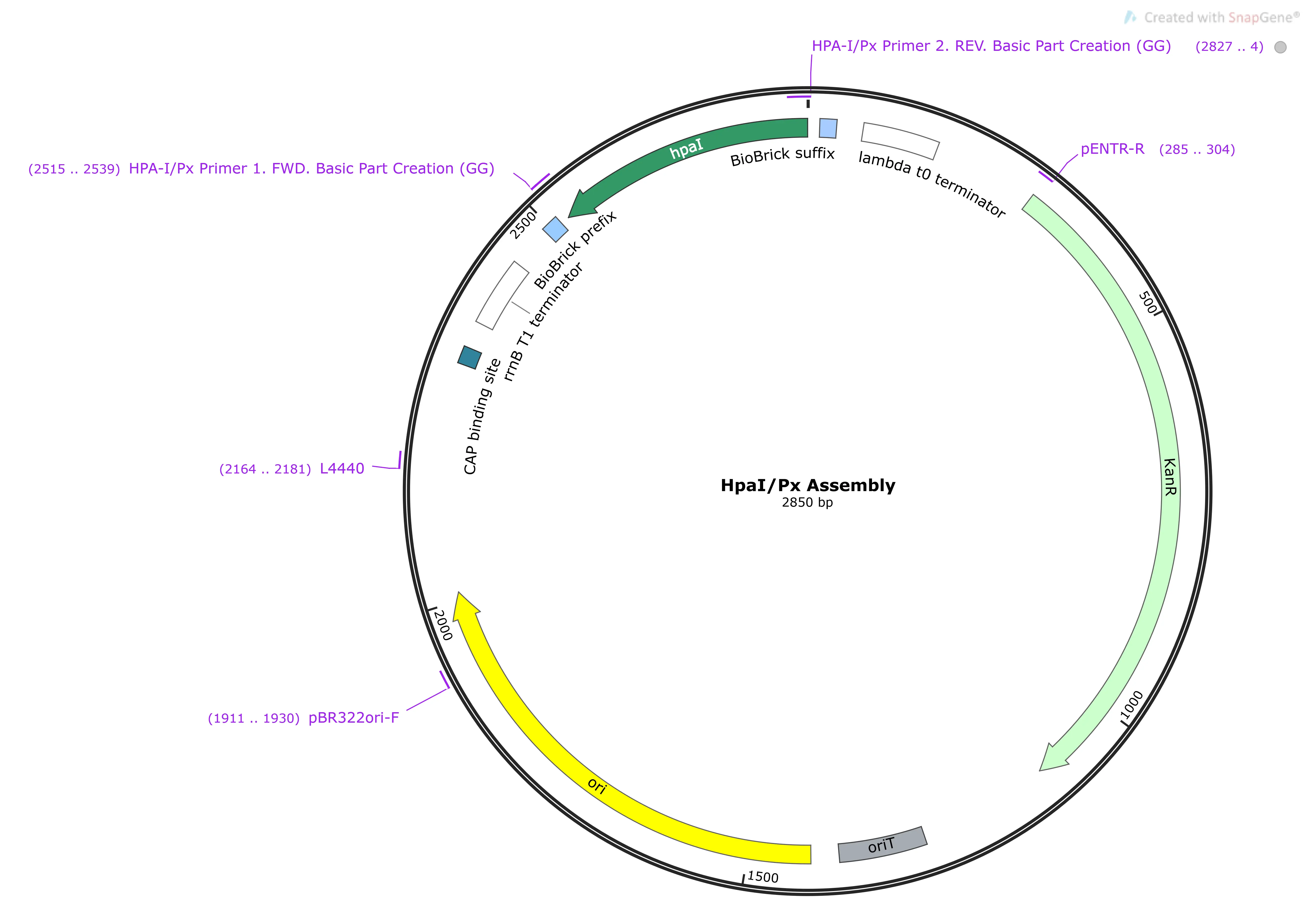
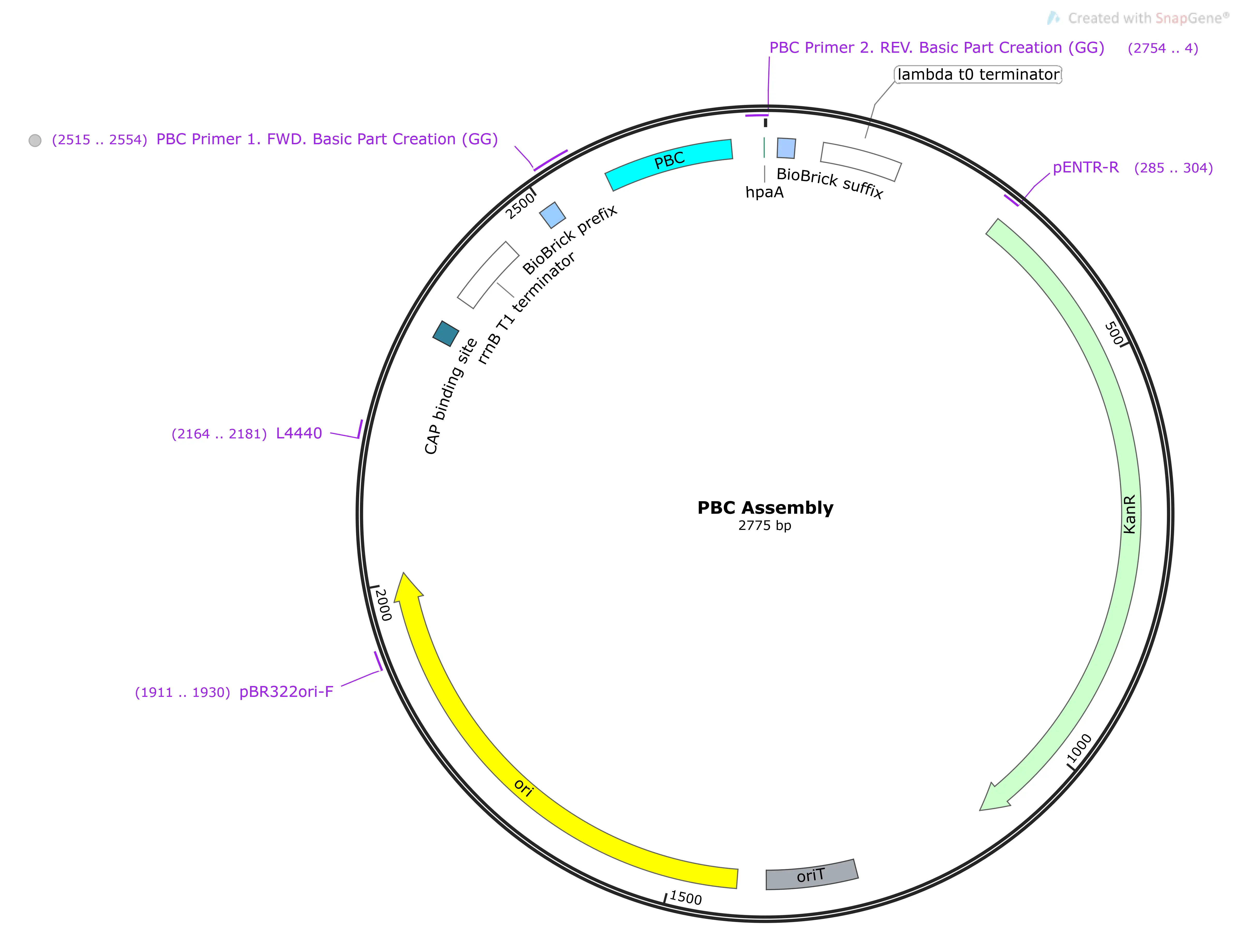
Pbc/PBC
The native promoter region for HpaB (a downstream gene of HpaX and A.)2
Basic Parts: BMD
BamD, A, B, C, and D
Bacillomycin D is a well characterized member of the Iturinic Lipopeptide family whose primary functions are antifungal in nature.3 In contrast, the function of each gene, Bam D, A, B, and C, are not well understood. There are speculative functions for each gene, however:
- BamD - biosynthesis malonyl-CoA transacylase
- BamA - hybrid PKS/NRPS
- BamB - non-ribosomal peptide synthetase
- BamC - non-ribosomal peptide synthetase4
We will not be including these parts in our wiki, as we did not work with these parts as intended, and the sequence data for these parts is convoluted and difficult to confirm in standard reference/well annotated strains.
Basic Parts: Plasmids
pJUMP 27-1A (altered from iGEM's distributed sequence)
“Joint Universal Modular Plasmids”5 is a system meant to optimize the Golden Gate cloning process. This collection can be found at Addgene, and in the standard registry of parts. Although many of the collections' plasmids are present in the iGEM 2022 distribution kit, the sequences do not align between the Registry and Addgene, at least for our plasmids. Due to part composition, we wanted to minimize domestication requirements. To do this we chose BsaI and AarI, corresponding to the Main and Upstream modules.

Composite Parts
Hpa Assembly
Our first composite part was created following golden gate assembly with BsaI.

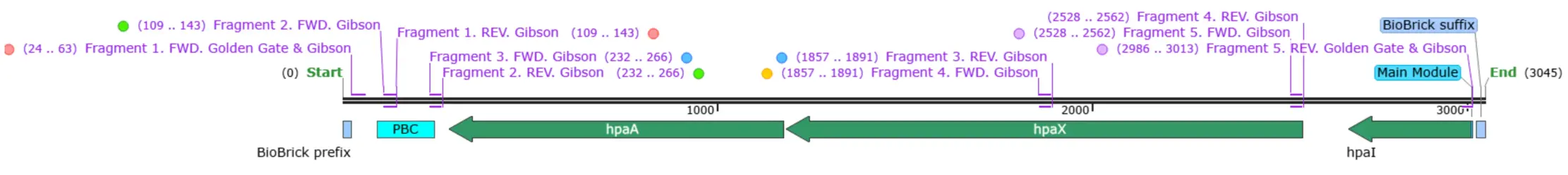
Hpa:GFP Assembly
Our second composite part was created following golden gate assembly with BsaI and AarI for Hpa and GFP respectively.
Downloads
| Part name | FASTA file | SnapGene DNA file |
|---|---|---|
| HpaA Assembly | HpaA Assembly.fa | HpaA Assembly.dna |
| Hpa Circuit Assembly | Hpa Circuit Assembly.fa | Hpa Circuit Assembly.dna |
| HpaI_Px Assembly | HpaI_Px Assembly.fa | HpaI_Px Assembly.dna |
| HpaX Assembly | HpaX Assembly.fa | HpaX Assembly.dna |
| PBC Assembly | PBC Assembly.fa | PBC Assembly.dna |
| pJUMP27-1A(sfGFP) | pJUMP27-1A(sfGFP).fa | pJUMP27-1A(sfGFP).dna |
| pJUMP28-1A(sfGFP) | pJUMP28-1A(sfGFP).fa | pJUMP28-1A(sfGFP).dna |
Citations
- Prieto, M. A., Díaz, E., & García, J. L. (1996, January). Molecular characterization of the 4-hydroxyphenylacetate catabolic pathway of Escherichia coli W: engineering a mobile aromatic degradative cluster. Journal of Bacteriology, 178(1), 111-120.
- Tscherner, M., Giessen, T. W., Markey, L., Kumamoto, C. A., & Silver, P. A. (2019, January 4). A Synthetic System That Senses Candida albicans and Inhibits Virulence Factors. ACS Synthetic Biology, 8(2), 434-444. https://doi.org/10.1021/acssynbio.8b00457
- Jin, P., Wang, H., Tan, Z., Xuan, Z., Dahar, G. Y., Li, Q. X., Miao, W., & Liu, W. (2020, February). Antifungal mechanism of bacillomycin D from Bacillus velezensis HN-2 against Colletotrichum gloeosporioides Penz. Pesticide Biochemistry and Physiology, 163, 102-107. https://doi.org/10.1016/j.pestbp.2019.11.004
- Moyne, A. L., Cleveland, T. E., & Tuzun, S. (2004, May). Molecular characterization and analysis of the operon encoding the antifungal lipopeptide bacillomycin D. FEMS Microbiology Letters, 234(1), 43-49. https://doi.org/10.1111/j.1574-6968.2004.tb09511.x
- Valenzuela-Ortega, M., & French, C. (2021, January 1). Joint universal modular plasmids (JUMP): a flexible vector platform for synthetic biology. Synthetic Biology, 6(1). https://doi.org/10.1093/synbio/ysab003