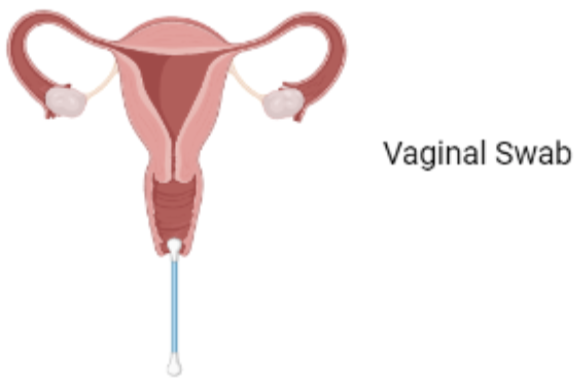
Step1: Sample Collection
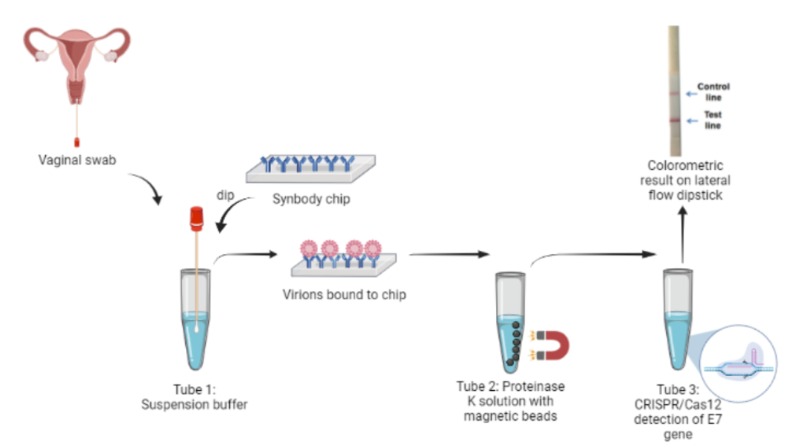
The first step is the collection of a viral sample from the site of infection. The test taker is required to collect a vaginal swab as opposed to a traditional cervical swab. A cervical swab such as the Papanicolau smear is an invasive sampling method which may compromise participation. Vaginal swabs have been shown to have viral loads comparable to cervical swabs and were able to identify a larger number of viral genotypes. [1] Thus, a shift from cervical swabs to vaginal swabs provides an opportunity for much wider screening coverage. Other sites of infection such as the anus, penis and the mouth can also be used for sample collection.
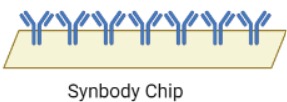
Step2: Virus enrichment
The collected sample is immersed in a suspension buffer to resuspend the viral particles. To increase the efficacy of the test, a synbody chip is used to purify the viral particles from the rest of the sample. Synbodies, or synthetic antibodies are bivalent peptides with a high affinity and specificity that strongly bind to specific target proteins for capture.The HPV viral capsid is primarily composed of the L1 and L2 virally encoded proteins. Of these, L1 protein is the major component of the capsid which mediates cell surface binding. Cell-surface Heparan Sulfate Proteoglycans (HSPGs), mainly syndecans and glypicans, are the primary receptors in the human body for the virus. They are complex molecules composed of a core protein with covalently attached glycosaminoglycans chains, but they can’t be used for viral purification from the sample as they are also the primary host cell receptors for many other viruses such as HSV-1, Dengue virus and Echovirus 5.[2] The HPV virion also associates with receptors such as alpha integrins, laminins, and annexin A2.[3.] Thus, the chip would consist of two L1-specific peptides, Alpha integrin 6 and Annexin A2, bound to an orthogonal scaffold group attached to a nitrocellulose base. This chip, when dipped in the resuspended sample, would immobilize the virions and allow them to be exclusively transferred.

Step3: DNA extraction
The chip is deposited in a solution containing Proteinase K in order to degrade the capsid and release viral DNA. Proteinase K is a fungal-derived enzyme capable of degrading a wide range of proteins and is commonly used during nucleic acid purification. Activated by Ca2+, the enzyme predominantly cleaves peptide bonds adjacent to the carboxyl group of aliphatic and aromatic amino acids with blocked alpha amino groups. The viral proteins L1 and L2 are rapidly digested, releasing the DNA into the solution. Viral DNA is extracted using silica coated magnetic nanoparticles in the presence of a chaotropic salt such as GuSCN, which facilitates DNA-silica binding. Lysate is added to the solution and particles are aggregated at the bottom of the tube using an external magnet. The supernatant is removed, followed by the addition of an elution buffer which solubilizes the DNA. The solution containing DNA is then used for further steps.
Step4: Amplification of E7 gene
The viral load present in the vaginal swab samples are subject to variation. To increase the sensitivity of our kit, isothermal recombinase polymerase amplification (RPA) is proposed to amplify the target sequence. RPA can be performed at 37℃ and has been shown to amplify as low as 1- 10 target DNA copies within 20 minutes. We chose this process as it would not require specialized equipment. CRISPR is a sensitive reaction and according to our literature review and interactions with a few CRISPR experts, we can eliminate this step. However, to confirm this hypothesis, we would have to actually do this in the lab which is beyond the scope of this project
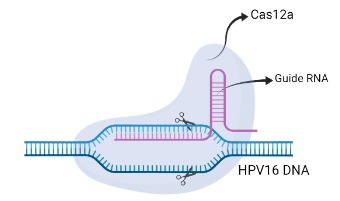
Step5: CRISPR/Cas12a
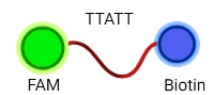
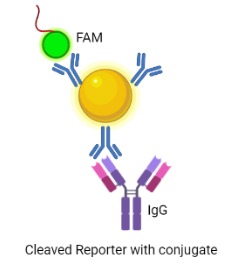
Cas 12 is a Class 2 CRISPR-associated protein with the property of trans-cleavage, i.e. nonspecific cleavage of ssDNA. This attribute is only activated once bound to an activator (target ssDNA) that has complementary base-pairing to the guide crRNA. Recombinant LbCas12a (from Lachnospiraceae bacterium ND2006) would form a ribonucleoprotein (RNP) complex with the crRNA and the target DNA, enabling the cleavage of ssDNA reporters in the reaction mixture.The chosen target is a region of the HPV16 E7 gene, found to be highly conserved among all HPV16 strain variants.[4] The ssDNA probe used as the reporter is FAM-TTATT-Biotin, as LbCas12a cleaves at AT rich sequences. Presence of the E7 gene in the sample would result in reporter cleavage and separation of a fluorophore, FAM (Fluorescein amidites) at the 5’end from biotin at the 3’end. This allows for detection, qualitatively via FAM-antibody binding, or quantitatively via fluorescence measurements.
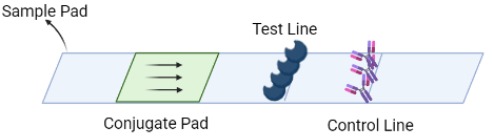
Step6: Visual Readout on LFA
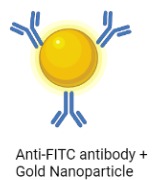
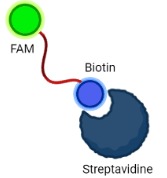
A Lateral Flow Assay (LFA) strip is employed for the visual readout of the test result. The strip consists of a sample pad on which 40 μl of the solution is dropped. Capillary forces drive the solution toward the conjugate pad, which consists of anti-FITC antibodies bound to gold nanoparticles. Anti-FITC antibodies selectively bind and form a conjugate with the 5’ FAM of the reporter. The test line consists of streptavidin, a biotin ligand whereas the control line contains IgG antibodies, which bind to a FAM-anti FITC antibody conjugates.
If the sample contains no HPV target DNA, the solution would contain only uncleaved reporter molecules. The reporter-antibody conjugate bound to gold nanoparticles would interact with streptavidin and on immobilization would provide a coloured line. The control line would not be visible as all the anti-FITC antibodies would be bound to an uncleaved reporter and immobilized at the test line. Hence, presence of test line and absence of control line indicates a negative result. In the case of a sample positive for HPV DNA, the solution would contain cleaved reporter molecules, allowing for independent flow of 5’FAM and 3’ Biotin. FAM-Anti-FITC antibody conjugates bind to IgG at the control line and biotin binds at the test line. Hence, presence of both test and control lines indicates a positive result.
Complete Workflow
References:
- Coorevits, L., Traen, A., Bingé, L., Van Dorpe, J., Praet, M., Boelens, J. and Padalko, E., 2018. Are vaginal swabs comparable to cervical smears for human papillomavirus DNA testing?. Journal of Gynecologic Oncology, 29(1).
- Cagno, V., Tseligka, E.D., Jones, S.T. and Tapparel, C., 2019. Heparan sulfate proteoglycans and viral attachment: true receptors or adaptation bias?. Viruses, 11(7), p.596.
- Taylor, J.R., Skeate, J.G. and Kast, W.M., 2018. Annexin A2 in virus infection. Frontiers in Microbiology, 9, p.2954.
- Yoon, C.S., Kim, K.D., Park, S.N. and Cheong, S.W., 2001. α6 integrin is the main receptor of human papillomavirus type 16 VLP. Biochemical and biophysical research communications, 283(3), pp.668-673.
- Burk, R.D., Harari, A. and Chen, Z., 2013. Human papillomavirus genome variants. Virology, 445(1-2), pp.232-243.









